Bernard Thisse, PhD
Molecular control of embryonic development
How do you build a vertebrate embryo from naive pluripotent embryonic cells? We recently discovered that a complete embryonic axis, containing all tissues and organs of a normal embryo can be organized by instructing uncommitted pluripotent zebrafish embryonic cells with as little as two opposing gradients of morphogens, BMP and Nodal. Because the signaling pathways controlling early embryonic development have been conserved across evolution we predict that these finding can be extrapolated to the organization of early mammalian embryos. Therefore we are now trying to take control of aggregates of mouse embryonic stem cells (embryoid bodies), instructing them through experimentally engineered, spatially defined, morphogen gradients that should control fate and behavior of these pluripotent cells. Our ultimate goal is to use this approach to generate in vitro functional tissues and organs for future application in Regenerative Medicine.
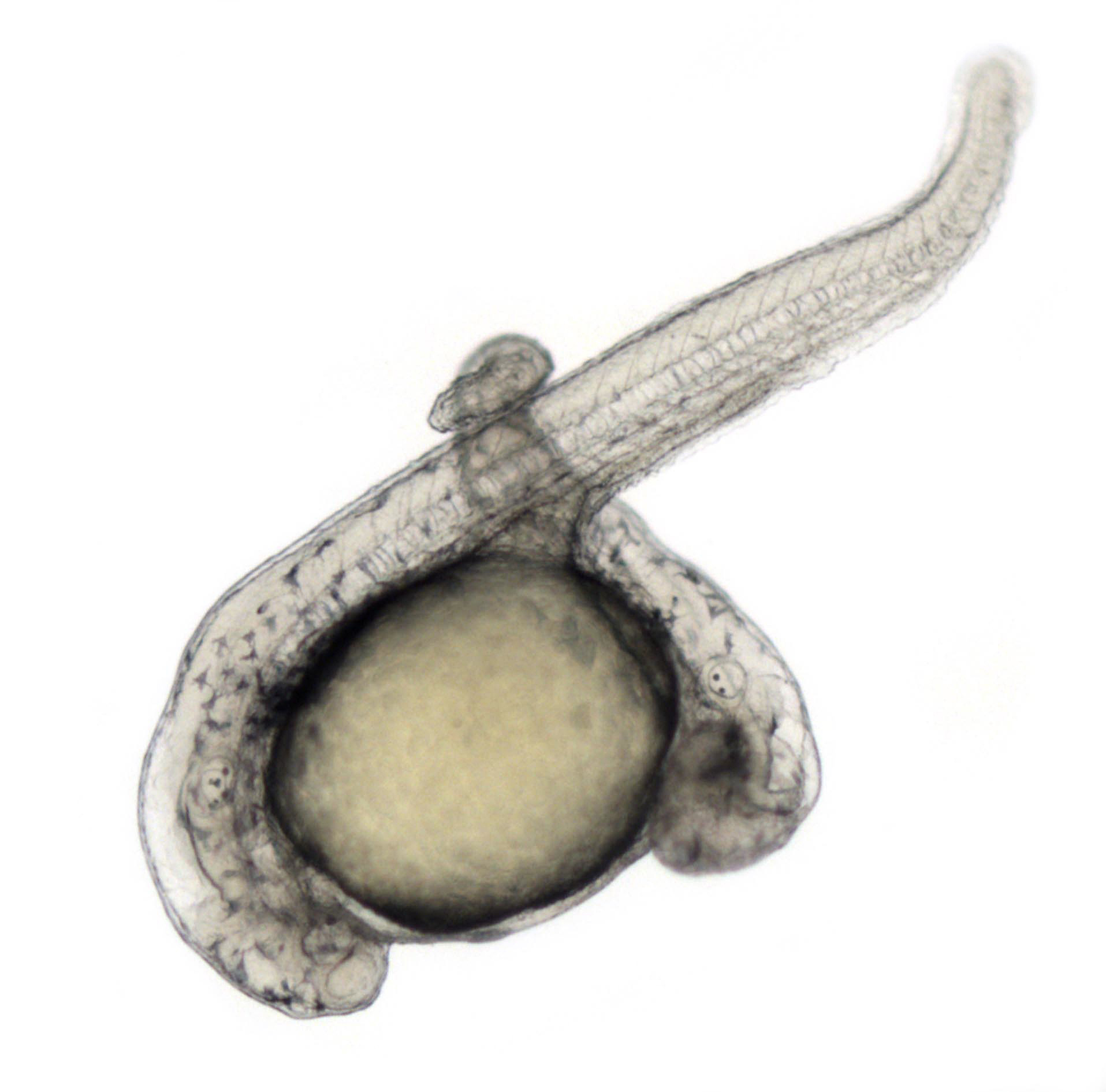
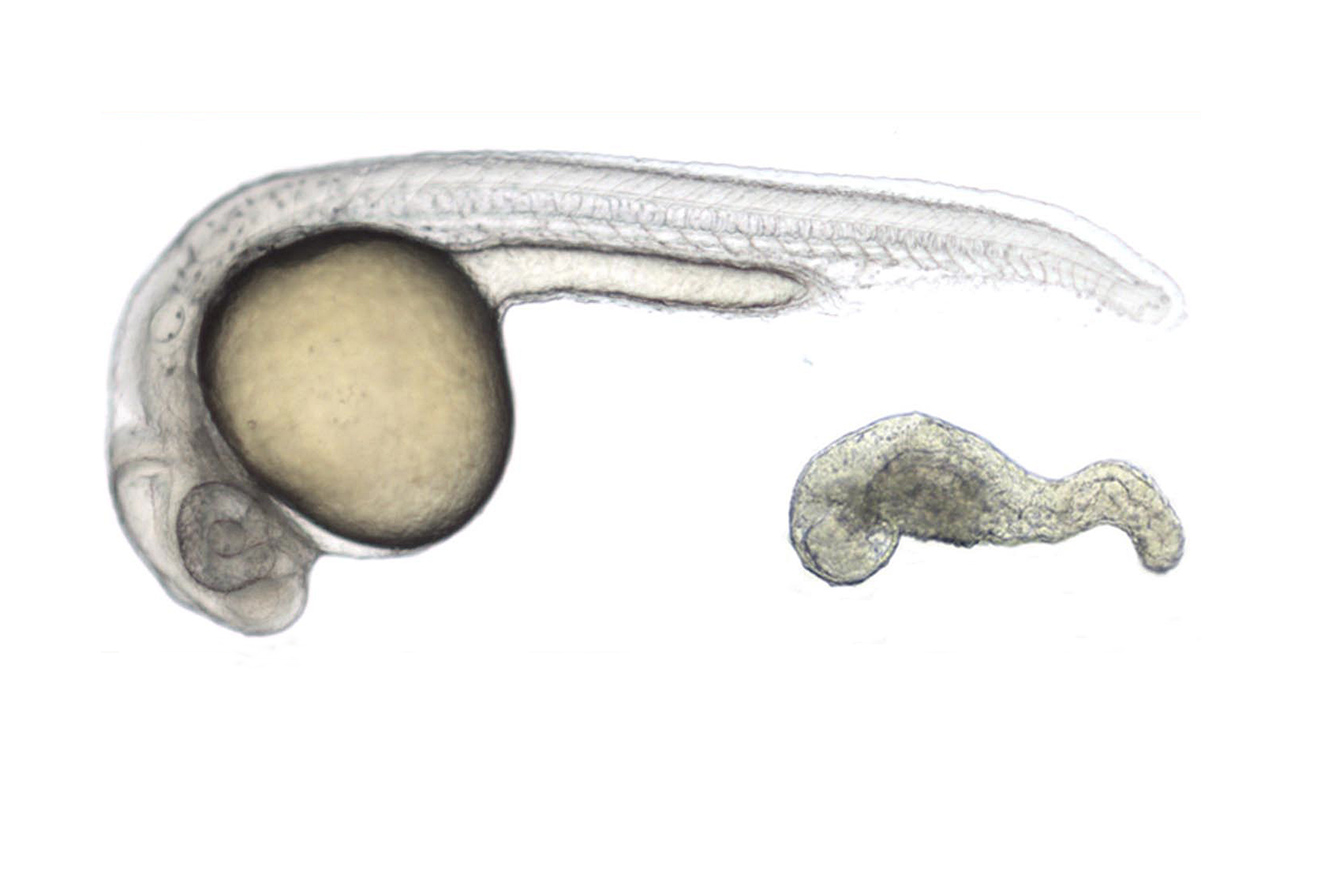
During the past two decades, our lab has dissected the network of molecular activities controlling the early embryonic development, focusing on the establishment and regulation of the formation of the dorso-ventral and antero-posterior axes. We are now studying how Left-Right asymmetry of the embryo is setup and controlled. We recently discovered an unsuspected role for factors mediating or regulating the transcriptional response of the Hippo signaling in the formation of the ciliated organ responsible for the Left-Right asymmetry: the Left-Right organizer. Using Crispr/Cas9 editing technology as well as a variety of gain of function and gene knockdown methods, we are currently dissecting the molecular mechanisms regulating the formation of this organ and through a comprehensive analysis of the transcriptome of its precursor cells, we will characterize the cascade of gene products involved in its formation and function.

